BREAKING: Largest Real-World Analysis of Ivermectin + Mebendazole in Cancer Patients Shows 84.4% Clinical Benefit — Nearly HALF Report Cancer Disappearance or Regression
After just 6 months, 48.4% of cancer patients taking ivermectin and mebendazole reported no evidence of disease (32.8%) or tumor regression (15.6%), while 36.1% reported disease stabilization.
We have completed the largest real-world human analysis to date evaluating ivermectin and mebendazole in cancer patients—and the results represent one of the most compelling clinical signals ever documented for repurposed anti-parasitic therapies in oncology.
The manuscript is now available as a preprint on the Zenodo research repository, operated by the European Organization for Nuclear Research, while undergoing peer review at leading oncology journals: “Real-World Clinical Outcomes of Ivermectin and Mebendazole in Cancer Patients: Results from a Prospective Observational Cohort.”
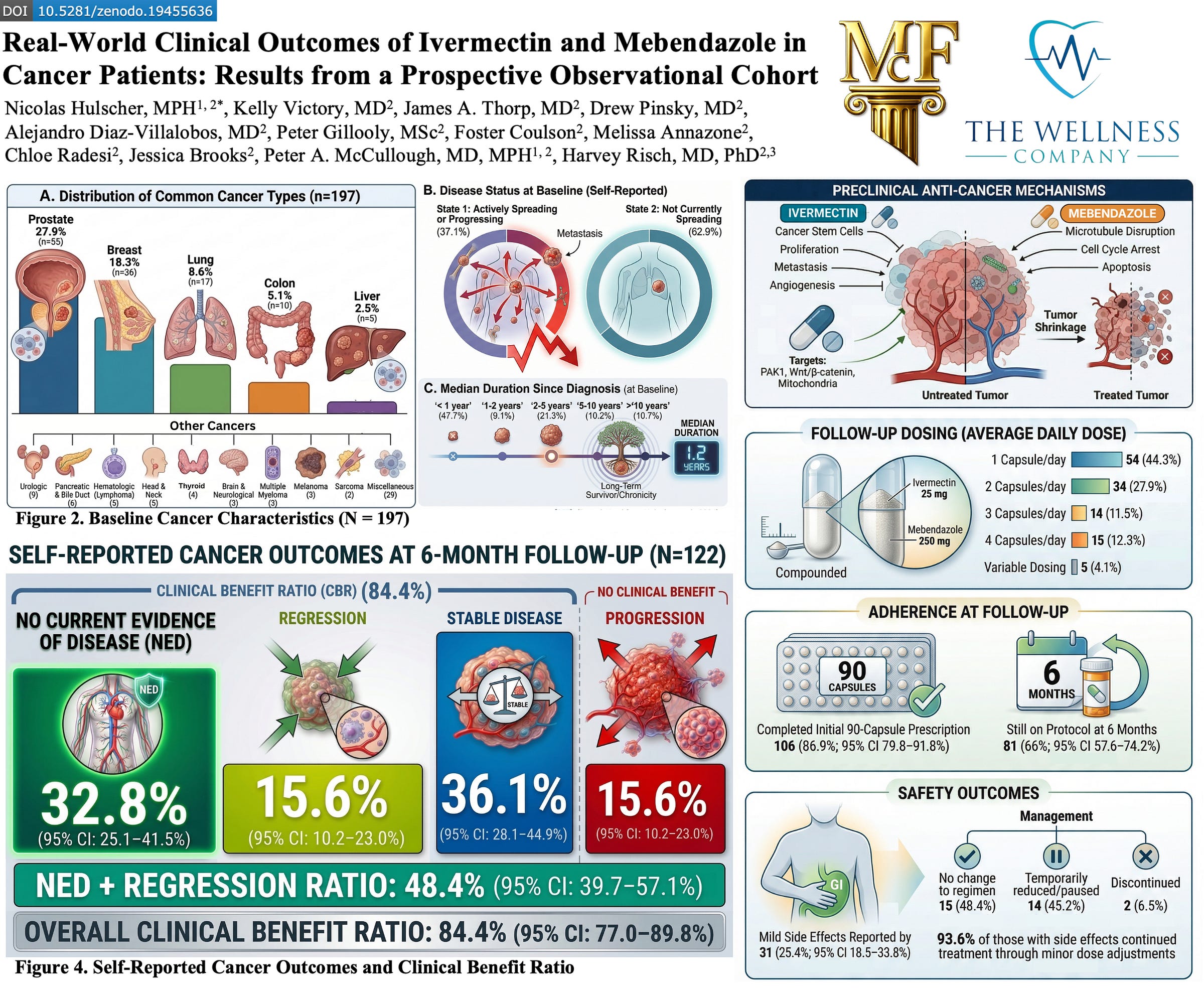
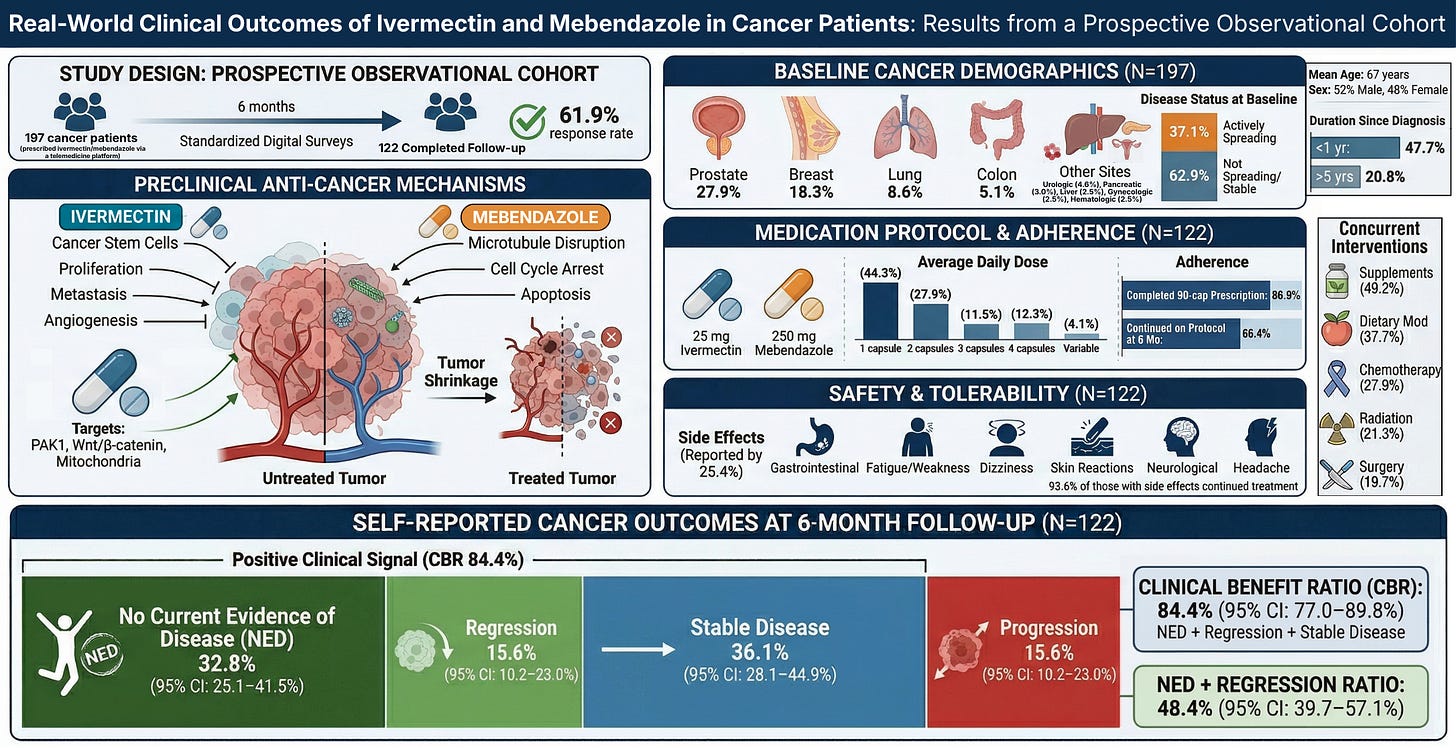
In this real-world prospective clinical program evaluation, a diverse population of cancer patients (n=197) were prescribed compounded ivermectin–mebendazole, with each capsule containing 25 mg ivermectin and 250 mg mebendazole.
At approximately six months post-treatment initiation, we observed an 84.4% Clinical Benefit Ratio (CBR), with nearly half of cancer patients (48.4%) reporting either no evidence of disease (32.8%) or tumor regression (15.6%). An additional 36.1% reported disease stabilization. This means more than four out of five patients reported either improvement or stabilization of their cancer.
These results indicate that the inexpensive and safe off-label applications of these medications could be an important complement in the treatment of cancer.
The groundbreaking analysis was made possible through a unique collaboration between The Wellness Company, the McCullough Foundation, and the Chairman of the President’s Cancer Panel—uniting real-world clinical data, frontline medical experience, and high-level epidemiologic expertise to deliver urgently needed insights in oncology.
This work was conducted by Nicolas Hulscher, MPH (myself); Kelly Victory, MD; James A. Thorp, MD; Drew Pinsky, MD; Alejandro Diaz-Villalobos, MD; Peter Gillooly, MSc; Foster Coulson; Melissa Annazone; Chloe Radesi; Jessica Brooks; Peter A. McCullough, MD, MPH; and Harvey Risch, MD, PhD (Chairman of the President’s Cancer Panel).
The paper can be accessed here: https://zenodo.org/records/19455636
A full breakdown of the analysis is below:
PROJECT DESIGN: REAL-WORLD DATA, PROSPECTIVE FRAMEWORK
We analyzed a prospective observational cohort of 197 cancer patients, with 122 completing structured follow-up at approximately six months (61.9% response rate). Patients were prescribed a compounded ivermectin–mebendazole protocol by licensed U.S. providers, and outcomes were collected through standardized digital surveys assessing cancer status, adherence, and safety.
Each capsule contained 25 mg ivermectin and 250 mg mebendazole, with dosing individualized by clinicians—most commonly 1–2 capsules per day, though a subset of patients used higher daily dosing or cyclic regimens depending on disease status and tolerance.
Importantly, this was a prospective, structured clinical program evaluation, capturing longitudinal patient-reported outcomes rather than retrospective recall alone—strengthening the internal consistency of the findings.
PATIENT POPULATION: ADVANCED, DIVERSE, AND CLINICALLY RELEVANT
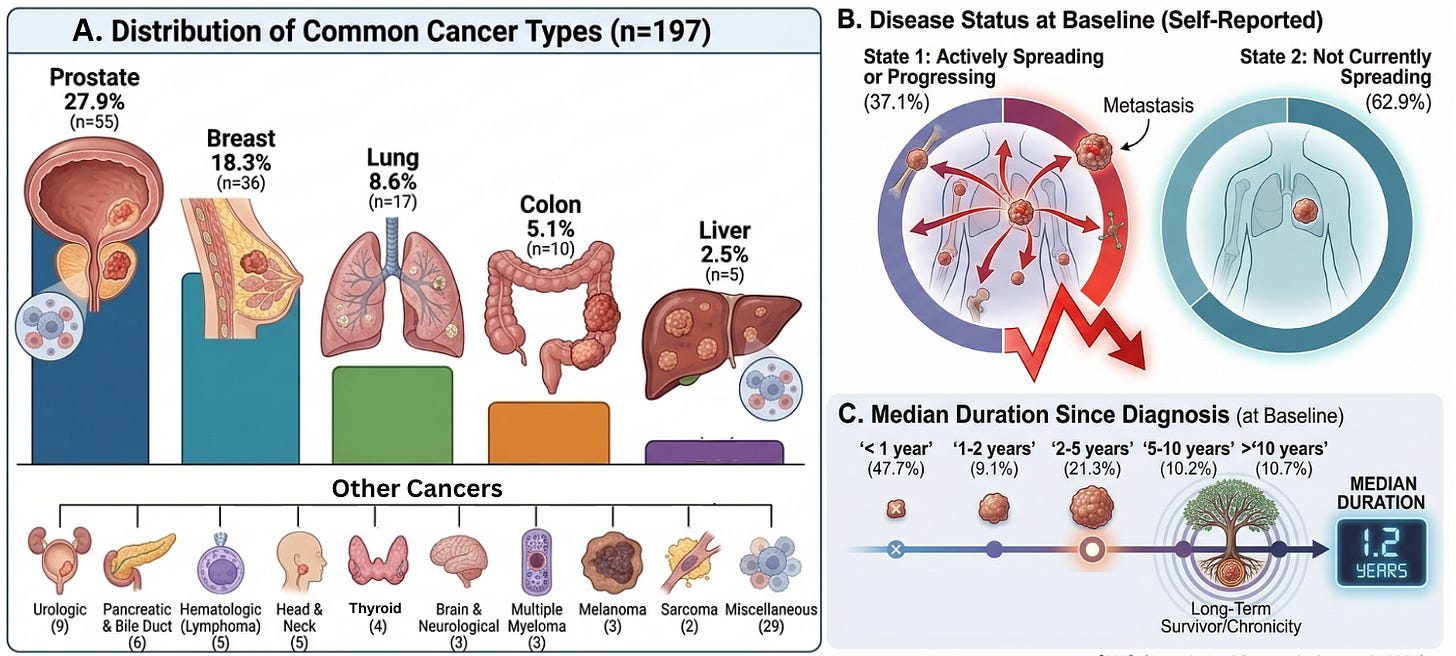
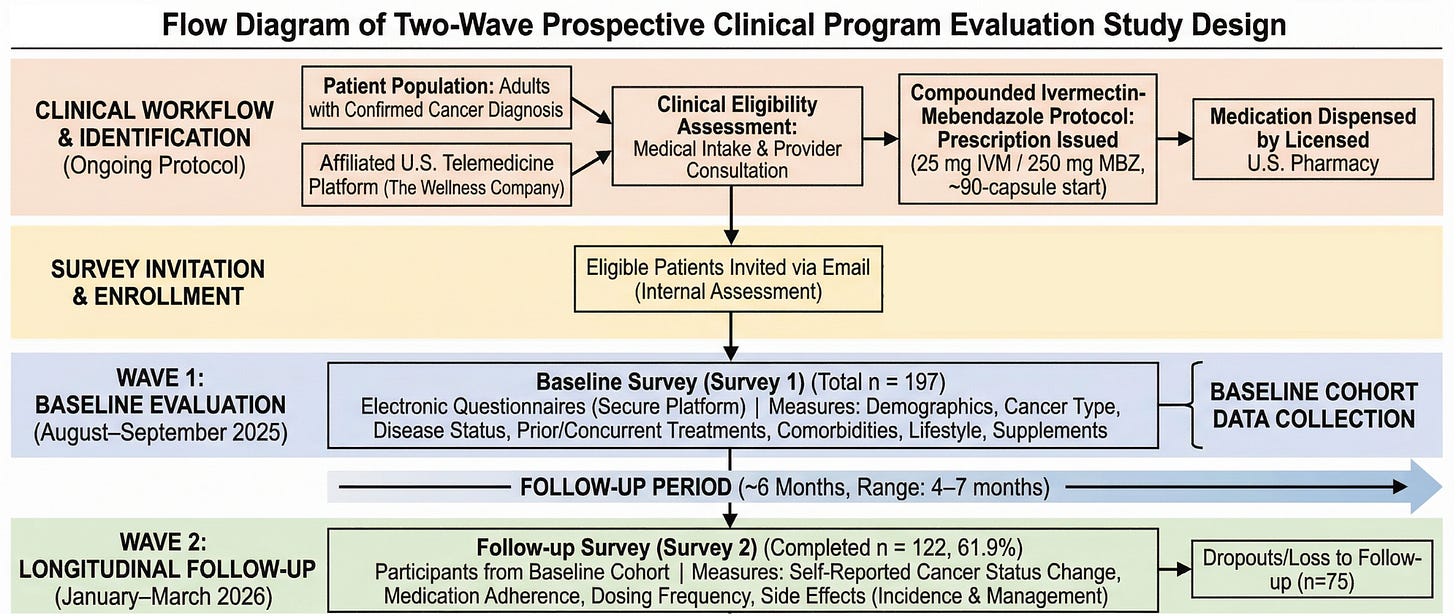
Our cohort represents a broad and clinically meaningful cross-section of cancer patients, including prostate (27.9%), breast (18.3%), lung (8.6%), colon (5.1%), and a wide range of additional malignancies.
This was not a population limited to early-stage or low-risk disease. At baseline:
- 37.1% of patients reported actively progressing cancer
- Nearly half were within one year of diagnosis, while others had long-standing disease
- Many had already undergone standard therapies:
- Chemotherapy (31.5%)
- Radiation (28.9%)
- Surgery (42.1%)
This reflects a real-world oncology population, including patients with treatment exposure, ongoing progression, and complex clinical histories.
PRIMARY OUTCOMES
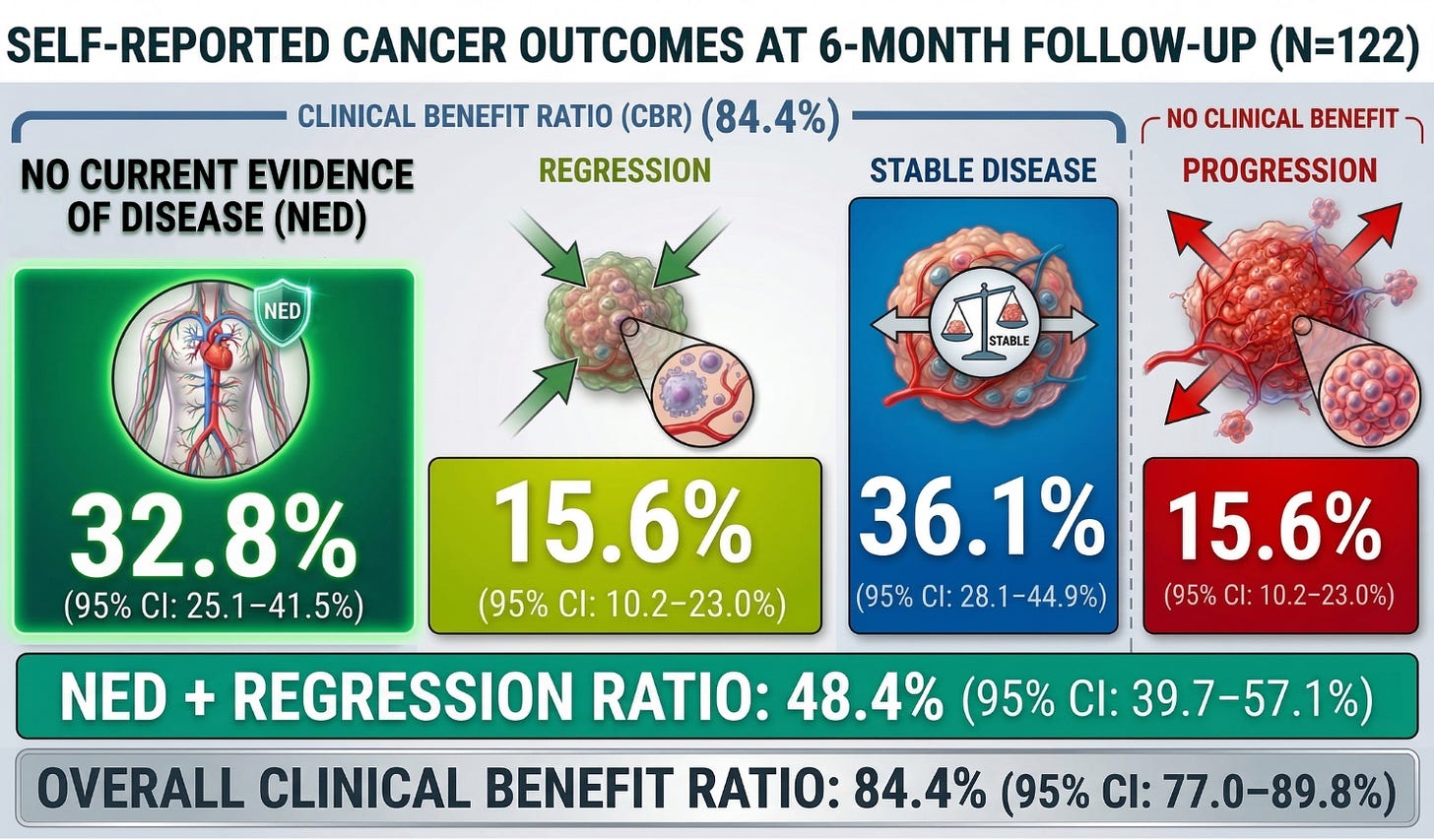
At approximately six months, outcomes were distributed as follows:
- No Evidence of Disease (NED): 32.8% (95% CI: 25.1–41.5%)
- Tumor Regression: 15.6% (95% CI: 10.2–23.0%)
- Stable Disease: 36.1% (95% CI: 28.1–44.9%)
- Progression: 15.6% (95% CI: 10.2–23.0%)
This yields a Clinical Benefit Ratio (CBR) of 84.4% (95% CI: 77.0–89.8%), meaning more than four out of five patients reported either improvement or stabilization of their cancer.
Critically, the 48.4% rate of NED + regression (95% CI: 39.7–57.1%) represents the strongest signal—indicating that a substantial proportion of patients did not merely stabilize, but reported meaningful reversal of disease burden.
ADHERENCE AND REAL-WORLD FEASIBILITY
Treatment adherence was notably high:
- 86.9% completed the full initial 90-capsule protocol
- 66.4% remained on therapy at six months
This level of adherence is uncommon in oncology and suggests that the regimen is both tolerable and acceptable to patients over extended periods.
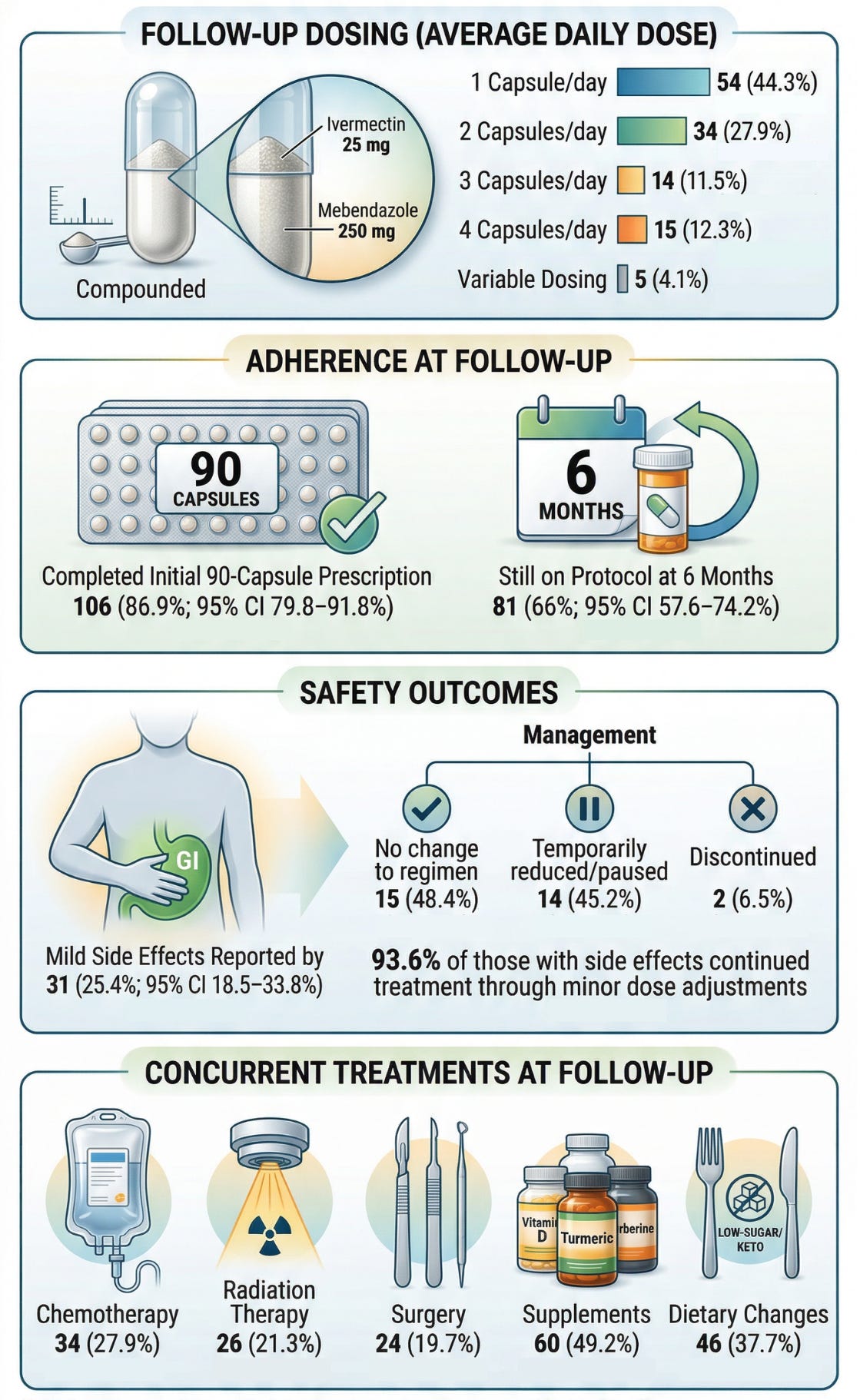
In addition, a many patients continued to receive concurrent therapies at follow-up, including chemotherapy (27.9%), radiation therapy (21.3%), and surgery (19.7%), alongside supplement use (49.2%) and dietary modification (37.7%).
Thus, the striking cancer outcomes were observed in real-world clinical conditions, where patients are often managed with multi-modality approaches rather than isolated monotherapy. The consistent signal of benefit in this setting supports the role of ivermectin and mebendazole as adjunctive therapies, capable of being integrated alongside standard-of-care treatments.
SAFETY PROFILE: LOW TOXICITY, HIGH CONTINUATION
Safety outcomes further support feasibility:
- 25.4% reported side effects, predominantly mild (e.g., gastrointestinal symptoms)
- 93.6% of individuals with side effects continued treatment with minor adjustments
This contrasts sharply with the toxicity burden of many conventional oncology therapies and supports the characterization of this protocol as a low-toxicity therapeutic approach suitable for long-term use.
CONCLUSION
This first-ever real-world analysis of a ivermectin–mebendazole protocol in human cancer patients provides a compelling signal that demands serious attention. While these findings should be interpreted appropriately as hypothesis-generating evidence from a real-world clinical evaluation, the magnitude, internal consistency, and broad distribution of the observed effects cannot be ignored. We are not observing marginal changes or isolated responses—we are observing widespread self-reported disease control across a diverse cancer population, a substantial proportion of patients reporting complete disappearance of detectable cancer, and sustained adherence with favorable tolerability over time.
Taken together, these results challenge the long-standing assumption that meaningful cancer responses must come exclusively from high-cost, high-toxicity therapeutic approaches. A signal of this magnitude—approaching 50% regression or no evidence of disease in a real-world population—would typically trigger immediate large-scale clinical investment if it originated from a novel, patent-protected pharmaceutical agent. Instead, these findings involve repurposed, low-cost drugs that have existed for decades, raising a fundamental question: how many clinically meaningful signals have been overlooked, deprioritized, or never pursued because they fall outside the conventional commercial drug development model?
This analysis does not close the case—but it decisively opens it in a way that can no longer be dismissed. The implications are clear. Prospective, randomized controlled trials are urgently needed to validate these findings, define optimal treatment strategies, and determine the full clinical potential of this protocol. Given the strength of the signal observed here, advancing this line of investigation is no longer optional—it is necessary.
This is not the end. We will continue advancing this work with larger datasets to further define and validate the role of anti-parasitics in cancer outcomes.
Epidemiologist and Foundation Administrator, McCullough Foundation
Support our mission: mcculloughfnd.org
Please consider following both the McCullough Foundation and my personal accounton X (formerly Twitter) for further content.




Apr 7
Very encouraging and very similar to what we found over a longer time period. Makis, W., Baghli, I., & Martinez, P. (2025). Fenbendazole as an Anticancer Agent? A Case Series of Self-Administration in Three Patients. Case reports in oncology, 18(1), 856–863. https://doi.org/10.1159/000546362.
Laura Kasner
Apr 7
“….a fundamental question: how many clinically meaningful signals have been overlooked, deprioritized, or never pursued because they fall outside the conventional commercial drug development model?”
Tragically, far too many. 😢
Yet another crime against humanity.